The export of domestic epidemic prevention materials has been tightened suddenly.
08 Apr 2020

The global epidemic situation is accelerating and spreading, and the situation is undergoing major changes.
In just three months, the number of people diagnosed with new crowns in the world has exceeded 1 million. The world\'s anti-epidemic disease is entering the most tense and tragic stage, and even more violent storms are sweeping towards the United States.
As of this morning (April 5), the cumulative number of diagnoses in the United States has exceeded 300,000. It is currently the country with the most confirmed cases.
With the sharp increase in the number of diagnoses in the United States, as well as in Italy, Spain, and Britain ... There are at least three shortages in the world: notched masks, lack of ventilators, and lack of virus detection reagents.

Trump saw the business opportunity at a glance. He had launched the Cold War-era National Defense Production Law, requiring GM and Ford to produce ventilators immediately. Obviously, Trump is going to do a big job-mass production of ventilator! Masks are worth a few dollars, a ventilator is priced at least 25,000 US dollars, and the technical threshold is also high. The ventilator is indeed a shortcoming of the big country made in China.
With the spread of the New Coronary Pneumonia epidemic worldwide, the demand for masks and other anti-epidemic materials has rapidly increased. China ’s world-wide factory-made machines are fully operational and are providing masks, virus detection reagents, protective clothing, protective gloves, infrared thermometers and other anti-epidemic materials to the world
Since the beginning of this year, the main business orders of foreign trade companies have been bleak, and they have also joined the cross-border epidemic prevention material export team, and are doing or preparing to do a big job in foreign trade! You pull a group of friends, how many people in the foreign trade circle are flipping their masks, and start a cross-border export business.

Along with this, the export of domestic epidemic prevention materials was out of order, and chaos appeared. The symbolic event is: Shenzhen Yirui Bio, the new coronavirus detection reagent exported to Spain is questioned locally with low accuracy, according to the Internet, only 30%, although Yirui Bio responded that the sample was not followed strictly according to the operating instructions. The detection accuracy dropped, but a wave caused thousands of waves and the national team shot.
On March 31, the Ministry of Commerce, General Administration of Customs, and the State Administration of Drug Administration announced No. 5 of 2020 regarding the orderly development of the export of medical materials, so that foreign trade companies, cross-border enterprises, and manufacturing enterprises are or are preparing to export virus detection reagents, medical Mask anti-epidemic materials caught by surprise!
Notification requirements: Since April 1, companies exporting new coronavirus detection reagents, medical masks, medical protective clothing, ventilators, and infrared thermometers must provide written or electronic statements when they declare to the customs, promising that the exported products have obtained Chinese medical treatment. The device product registration certificate meets the quality standards of the importing country (region).

In short: Double certificate declaration, CFDA, CE can only be exported.
The epidemic situation in foreign countries is now fierce, and the supply of masks and testing reagents is extremely important. The epidemic prevention materials are strategic materials. Most of the world\'s epidemic prevention materials are mainly produced in China. The quality of medical device products is closely related to the responsibility and image of major powers.
The announcement on the export of medical materials of the Ministry of Commerce and the hot topic of the export quality of medical equipment products in the industry. Recently, the editor visited a well-known local new coronavirus detection reagent manufacturer in Xiamen-De Biqi Biotechnology (Xiamen) Co., Ltd., referred to as TBG, and interviewed Mr. Yang Jian, Vice Chairman of TBG.

Mr. Yang Jian, a northerner, accurately speaks from Shandong, as his name implies, talkative, bold, straightforward, straight to the topic, as soon as I met, I said, President Hu, you are the first person in the mining and foreign trade media, I saw yours The outbreak of the European and American epidemic on the media platform of Bao Rui De\'s official account broke out, and foreign trade orders bleed up. What are you doing?
Regarding the originality of the domestic new coronavirus detection reagents, the content is wonderful, the information is large, the spread is fast, the influence is large, foreign trade media people, exporters participate in the overseas marketing of anti-epidemic materials such as detection reagents, using your foreign trade big data, integrated marketing advantages and mature Foreign trade channels can fully achieve strong alliance.
Yang Dong emphasized that the current overseas epidemic situation is very urgent, and TBG is going all out to promote exports and help overseas epidemic prevention. He said that recently, the state issued regulations requiring double-certification (CFDA, CE) customs declaration. It is indeed not a bad thing to ensure product quality and regulate export order. At this point of time, it is considered very meaningful.
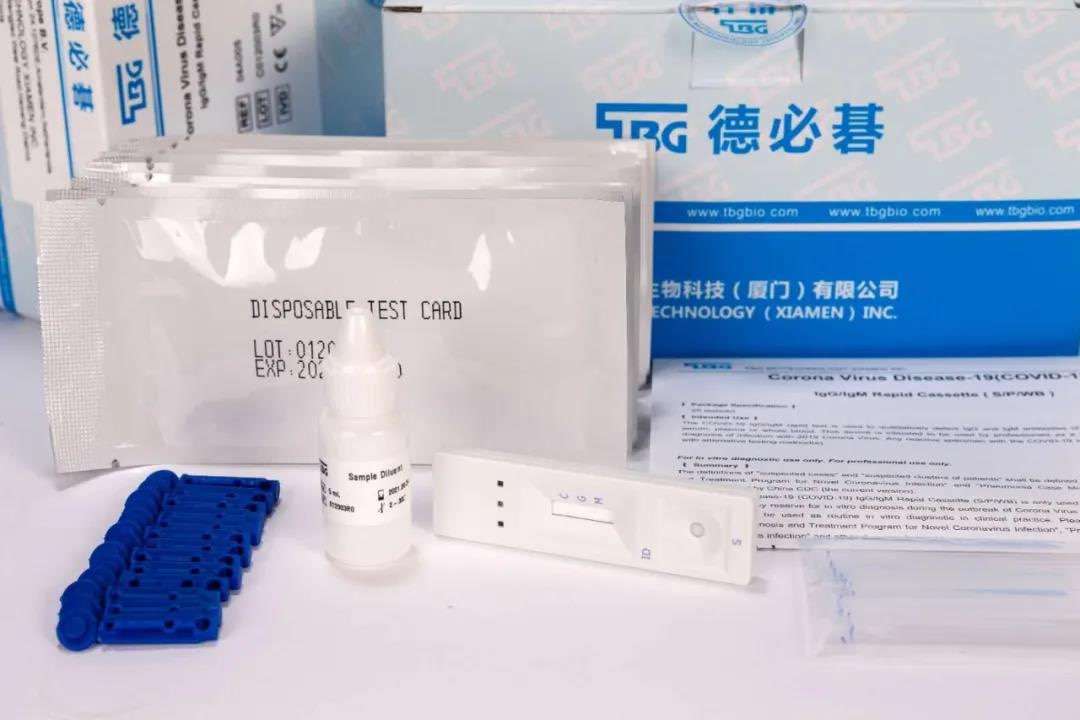
He said that in vitro testing reagents are classified as the lowest risk category in the EU, which is much lower than the three types of medical devices for Chinese entry barriers. The so-called registration is not a registration in the true sense, because it does not have a technical review, or Clinical verification, this link, just made a record.
In this way, a large number of companies produce related products. It is easy to get the record in the EU. Even if the products produced may not be qualified, they can still get the record and can still be exported to the EU.
In the final analysis, each national and regional medical device regulation is caused by different regulations, Yang Dong further explained.

According to the statistics of medical device product registration information published by the State Food and Drug Administration, there are 752 registered enterprises for disposable medical masks and 301 registered enterprises for medical protective clothing. Why are there only 23 registered enterprises for new crown virus detection reagents?
Indeed, there are relatively few new coronavirus detection reagents (enterprises), and Xiamen also has several kits that are not included in the registration list. This is related to the technical difficulty and clinical risk of the product.
Debiqi Hunan subsidiary, Changsha Nagano Medical Laboratory, under the leadership of the Hunan Provincial Health Commission, carried out the new coronavirus test work, and has passed the interim quality assessment of the New Coronavirus nucleic acid testing room of the National Health Commission.
In addition, the registration time of domestic medical device products can be as short as 4-5 months and as long as half a year.

Overseas, the circulation of medical devices in the United States requires FDA certification, but as new viruses and new medical devices, it is understood that no domestic company has passed the FDA.
Therefore, for many foreign trade enterprises that do cross-border anti-epidemic materials, regardless of whether they hold the EU CE certification or other certifications on hand, it may be more difficult to export in the short term.
Speaking of TBG\'s overseas exports, Yang Dong said that at present the company is carrying out certification work in full swing. The products are: new coronavirus (SARS-CoV-2) nucleic acid detection kit (fluorescent probe method), new coronavirus IgG / IgM antibody detection reagent.
The company has invested a lot of manpower, material resources, and financial resources in the new coronavirus detection reagent project. The company has strong technical force, standardized production, excellent quality, and system compliance.
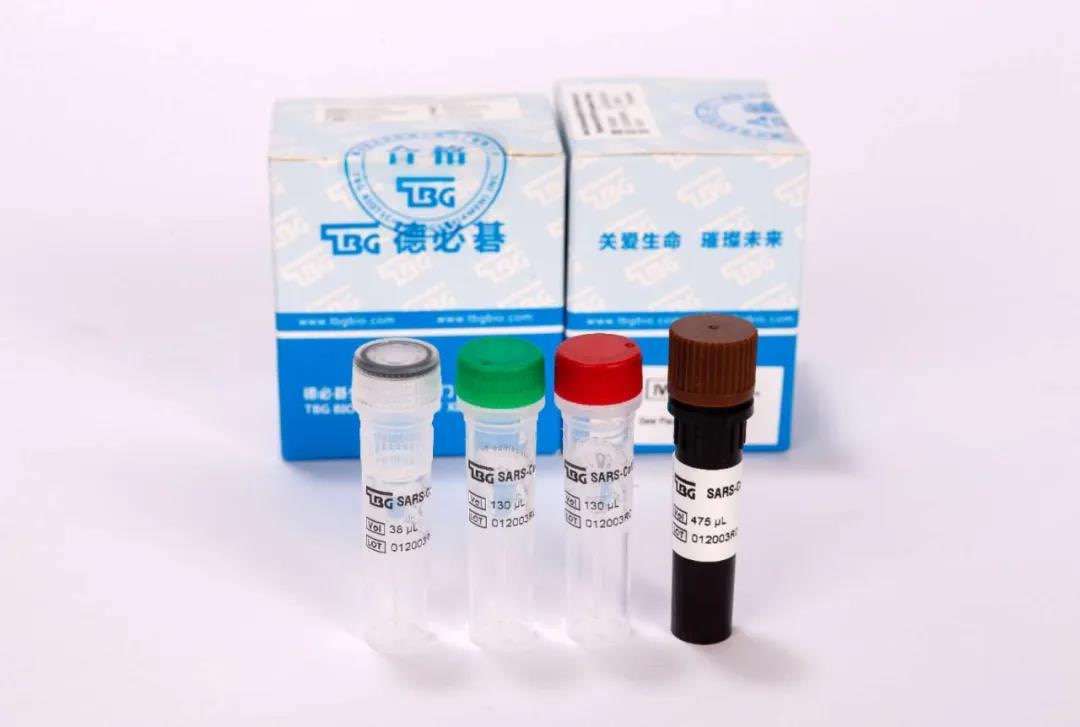
Pictured: Novel Coronavirus (SARS-CoV-2) Nucleic Acid Diagnostic Kit (PCR-Fluorescence Probing)
In addition, he said that unlike other domestic production companies, TBG is a global, international company. TBG\'s research and development bases are located in the United States and Taiwan. It is produced in mainland China and listed on the Australian Stock Exchange in 2016. Taking orders is a major advantage of our TBG.
At present, the export orders are produced and shipped from the factory in New Taipei City, Taiwan. Everything is in the process of intensive promotion. At the same time, OEM is also carried out in Southeast Asia. Later production will be greatly improved, and key raw materials are being deployed in the near future!

The first quarter of 2020 is over. This should be the most memorable, most anxious, most thrilling, and most ups and downs quarter in our lifetime.
At present, the global epidemic situation is accelerating and spreading, and there will be the second and third quarters. I hope this is not a long war, and don\'t continue to see the tragedy that is going on.
The play must start from the prologue, but the prologue is not yet a climax. The most miserable this year is the foreign trade white-collar workers; the more miserable than the foreign trade little white-collar workers is the foreign trade boss.

The epidemic has changed the world and China, and is changing the pattern of China\'s foreign trade. Crisis Crisis, crisis, organic, for China\'s foreign trade, this is a serious challenge, but hold it up, it is an opportunity.
Foreign traders in the turbulent waves must share the same frequency with the times, resonate with the city, and participate in the global anti-epidemic. Find the hope of life, the reason to live, live, and keep the roots.











